| University | Nanyang Technological University (NTU) |
| Subject | Thermodynamics |
Thermodynamics Project Questions 2026
Before starting to work on the questions, please DO read the information below.
If you have any questions about the project, please post them to the Q&A forum on Moodle.
Your answers to the questions must not be more than 12 pages of A4 size paper in total. If you are using a digital notebook, i.e., OneNote or similar, make sure to set the paper size to A4.
Handwritten work is permitted but must be legible.
Question 1
Shown below in Figure Q1 is a two-stage cascade refrigeration arrangement, using HFC-134a as the working fluid. The system comprises two ideal vapour-compression refrigeration cycles coupled through an internal heat exchanger: the condenser of the low-temperature (cold) cycle rejects heat to the evaporator of the high-temperature (warm) cycle. Given that the molar flow rate in the warm cycle is 0.50 mol s⁻¹, determine the following:
a) the molar flow rate through the cold cycle. [10]
b) the refrigeration capacity, i.e., the rate at which heat is extracted from the cooled space. [10]
c) total compressor power requirement (sum of the compressors in both cycles). [10]
d) the overall coefficient of performance (COP) of the cascade system. [5]
e) identify a more sustainable alternative to HFC-134a for use in vapour-compression refrigeration (state the refrigerant name). Then comment on the relative Global Warming Potential (GWP) / environmental impact of the alternative versus HFC-134a and the safety classification considerations (e.g., toxicity and flammability). [5]
Your answers must include the table/graph/diagram data you used to obtain thermodynamic data for HFC-134a
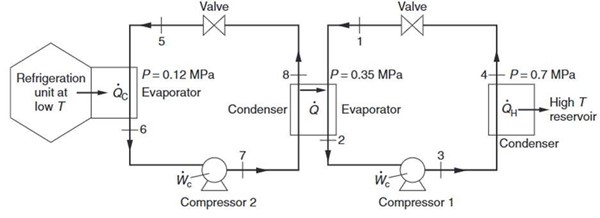
Figure Q1
Question 2
You have been tasked with developing a Rankine-cycle that delivers power at a rate of 10,000 kJ min-1. The turbine operates with an efficiency of 80 %, and the design places no constraints on the exit temperature, pressure, or vapour quality at the turbine outlet. Your objective is to select operating conditions that yield the lowest-cost Rankine cycle. The boiler may be specified to operate at a saturation temperature of 240 °C, 230 °C, or 220 °C, while the condenser may be designed for either 40 °C or 50 °C operation. The purchase costs of the boiler and condenser are to be estimated using the correlations provided in the Table Q2.
Heat supplied to the boiler is priced at £12 per GJ. Heat rejected in the condenser is assumed to have no associated cost or recovery value. The power cycle is expected to run continuously for 24 hours per day, 360 days each year.
a) For each possible Rankine-cycle configuration, evaluate the heat input ܳு and heat rejection ܳ, estimate the capital cost of the boiler and condenser from the provided correlations, and compute the annual expenditure for the required heat supply. [25]
b) Identify the preferred Rankine-cycle configuration, among the alternatives considered, that should be selected given an anticipated operating lifetime of five years. [5]
Table Q2 (In the correlations provided, ܥdenotes the cost (in £), and ܳ̇ is defined as the magnitude of the heat-transfer rate through boiler or condenser, expressed in kJ min-1.)
| Operating Temp (°C) | Formula | |
| Boiler | 220 | C = 10000 + ܳ̇ |
| Boiler | 230 | C = 12000 + 1.5ܳ̇ |
| Boiler | 240 | C = 15000 + 2ܳ̇ |
| Condenser | 40 | C = 10000 + 1.5ܳ̇ |
| Condenser | 50 | C = 7000 + ܳ̇ |
Question 3
A rigid 0.5 m³ vessel is linked via a valve to a piston–cylinder device, as shown in Figure Q3. Both containers hold pure water and are submerged in a constant-temperature bath at 200 °C and 600 kPa. Treat the combined tank + piston–cylinder as the system, and the constant-temperature bath as the surroundings.
At the start, the valve is shut, and both units are in equilibrium with the surroundings (the bath). The rigid tank initially contains saturated steam with a quality of 95 %. The piston–cylinder initially occupies 0.1 m³. The valve is then opened, allowing water to transfer into the piston–cylinder until a new equilibrium state is reached.
For this process, determine the entropy change of the system and the surroundings. [30]
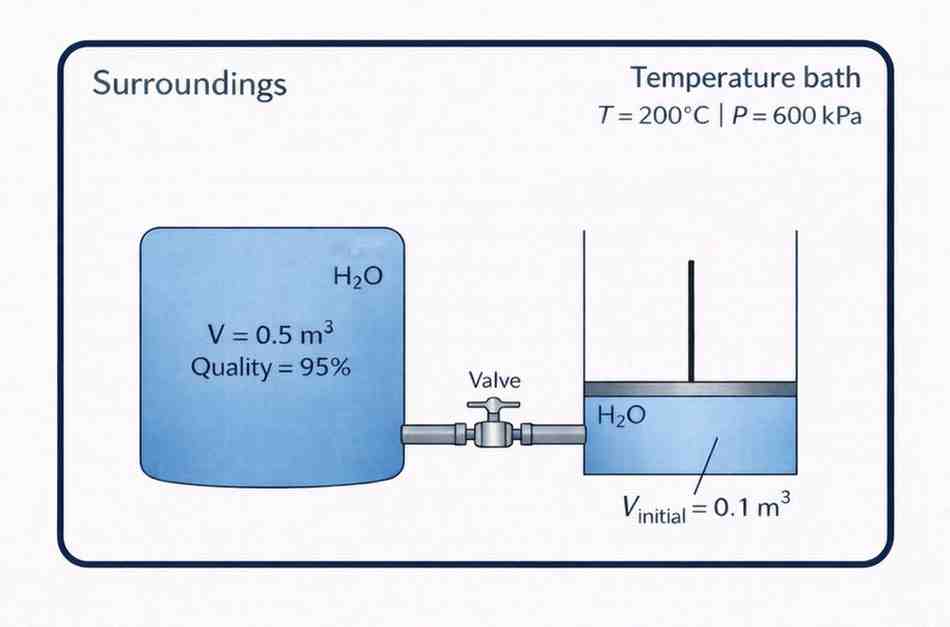
Figure Q3
Struggling with the Thermodynamics Project questions?
Native Singapore Writers Team
- 100% Plagiarism-Free Essay
- Highest Satisfaction Rate
- Free Revision
- On-Time Delivery
Many students in Ireland often find thermodynamics project questions difficult, especially when calculations involve cascade refrigeration systems, Rankine cycle analysis, and entropy balance problems. These projects require strong understanding of thermodynamic tables, engineering equations, and structured technical explanations within limited page requirements. If you are facing similar challenges, Singapore Assignment Help offers reliable Chemical Engineering assignment help designed according to university project guidelines. You can also review our expert-written engineering assignment solutions to understand the quality of work delivered by our specialists. If you need a properly explained 100% human-written thermodynamics project solution, you can hire our chemical engineering assignment helper today and receive a fully customised answer prepared specifically for your project.
Looking for Plagiarism free Answers for your college/ university Assignments.
- ICT302 Generative AI: Theory and Practice End-of-Course Assessment 2026 | SUSS
- GSS503 Navigating Risk in an Interconnected World Course Tutor-Marked Assignment 01, 2026
- GSS501 Global Crime Prevention and Security Management Tutor-Marked Assignment 01, 2026
- PSB6012CL Business Research Methods Assignment Brief 2026 | Coventry University
- MTH109 Calculus Tutor-Marked Assignment 1, 2026 | SUSS
- BUS286 Corporate Finance Assignment 2026 | Murdoch University
- HFSY359 Fatigue Management Tutor Mark Assignment Question 2026 | SUSS
- BSE313 Sport Coaching Tutor-Marked Assignment 2 Question 2026 | SUSS
- 6079MP Final Coursework Assignment 2026 | Coventry University
- IBUS2004 Managing International Business Assessment 1 Brief 2026 | UON

